Inspira’s pre clinical studies presentation
Initial Pre-Clinical Studies (In-vivo)
Dr. Revital Tuval, PhD
Forward Looking Statement
This presentation contains express or implied forward-looking statements pursuant to U.S. Federal securities laws. For example, the Company is using forward-looking statements when it discusses the conclusions from the preclinical studies on swine. These forward-looking statements and their implications are based on the current expectations of the management of the Company only and are subject to a number of factors and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Except as otherwise required by law, the Company undertakes no obligation to publicly release any revisions to these forward-looking statements to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events. More detailed information about the risks and uncertainties affecting the Company is contained under the heading “Risk Factors” in the Company’s Registration Statement on Form F-1 filed with the SEC, which is available on the SEC’s website, www.sec.gov.
Studies Results Highlights
- INSPIRA™ ART treatment in hypoxemic swine model, resulted in significant increase of oxygen saturation by 8%-10%
- A flow of 1 liter per minute, was sufficient to provide a saturation increase of 8-10%
- Pulmonary artery saturation increase is attributed to INSPIRA ART system activity. Documented saturation values in the Pulmonary artery represent INSPIRA ART activity as it captures oxygenation level just before the blood enters the lung. Additional increase in oxygen saturation may result via native lung gas exchange and is influenced by the lung condition and underlying lung disease.
- INSPIRA ART increased Pulmonary artery oxygen saturation by 26%. Increase was statistically significant
- INSPIRA ART exhibited a significant decrease of PaCO2. Decrease was statistically significant
Pre-clinical Studies (In-vivo)



The results are presented here from these studies
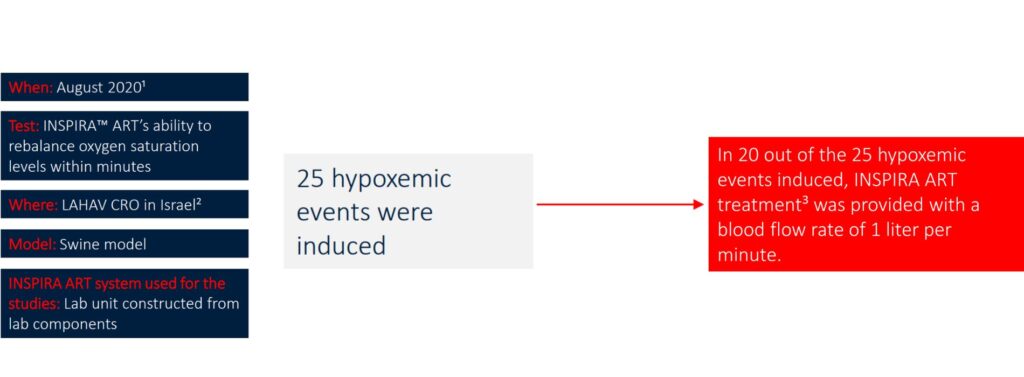
• During 2019-2020, additional 15 pre-clinical studies were conducted as feasibility tests.
• On November 2021, an additional pre-clinical study was conducted in LAHAV CRO in Israel.
1. Research protocol was approved by the national ethics committee of animal experimentations.
2. LAHAV CRO https://lahavcro.com/
3. INSPIRA ART Treatment = extracorporeal blood oxygenation with one liter of blood
Method
In these initial pre-clinical studiesthe goal was to assess the oxygenation effectiveness of INSPIRA™ ART system¹ in swine model.
Swine model
The swine species chosen for the current study is Large-White X Landrace. This breed was chosen due to a well-known resemblance of the anatomy, cardiovascular and respiratory physiology, size scale and other characteristics to adult humans.

Study setup
Two anesthetized mechanically ventilated swine, were cannulated in the right internal jugular vein via a double lumen cannula. The cannula conduits were connected inlet & outlet tubes, allowing for blood transportation to and from the veno-venous INSPIRA ART device.

Hypoxemia induction
Data Collected
Clinical and physiological parameters were collected throughout the test. Oxygenation was assessed by means of a non-invasive sensor (PPG-Sat), and blood gas measurements. Both arterial and venous blood were drawn and analyzed using the GEM 4000 Blood Gas Analyzer (Werfen, Barcelona, Spain). Blood pressure was assessed from pressure probes located in the femoral vein and carotid artery. An arterial sample (drawn from the carotid artery) and a venous sample (drawn from the femoral vein) were assessed for partial oxygen pressure (pO2 ), oxygen-bound hemoglobin (O2HGB), blood gas saturation, carbon dioxide partial pressure (pCO2), bicarbonate levels (HCO3), hemoglobin (HGB), hematocrit (HCT), pH, glucose, and electrolytes (sodium, potassium, chloride and ionized free calcium).
Study design


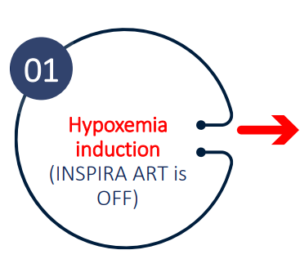
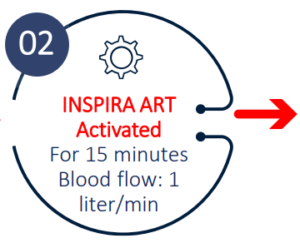
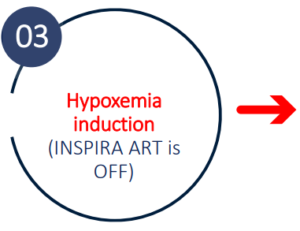

Study Results
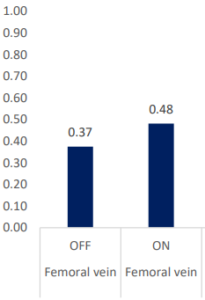
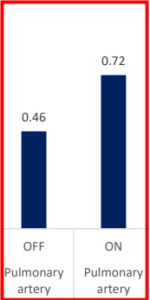
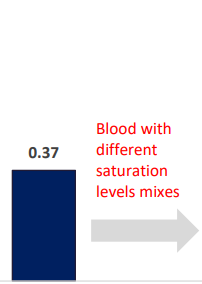
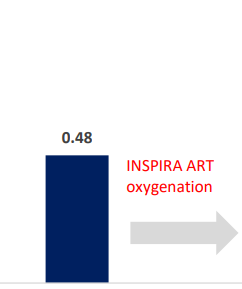
INSPIRA ART Provides Significant Increase In Saturation
SvO2 & SaO2_INSPIRA ART in OFF mode
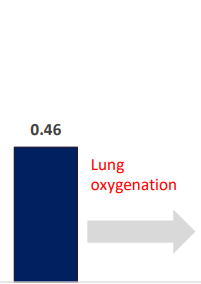

SvO2 & SaO2_INSPIRA ART in ON mode

| Parameter | Blood sample location | INSPIRA ART Status |
INSPIRA ART Status |
Difference | Std Error | t Ratio | Prob>|t| | Lower 95% | Upper 95% |
| SvO2 (%) | Femoral vein | OFF | ON | -0.106405 | 0.029932 | -3.55 | <0.005 | -0.1694 | -0.04341 |
| SaO2 (%) | Pulmonary artery | OFF | ON | -0.264375 | 0.034515 | -7.66 | <.0001 | -0.33689 | -0.19186 |
| SaO2 (%) | Carotid artery | OFF | ON | -0.080208 | 0.009376 | -8.55 | <.0001 | -0.09991 | -0.06051 |
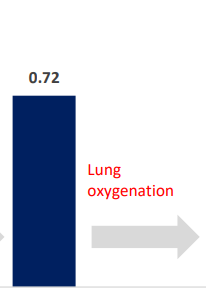
INSPIRA™ ART’s Contribution to Oxygen Saturation Should Be Reviewed Independently From the Lung Activity
INSPIRA ART increased saturation by 26% in the Pulmonary Artery
The contribution of INSPIRA ART to saturation should be assessed via saturation measured in the Pulmonary artery; right after ART oxygenates 1 liter of blood and just before the patients’ entire Cardiac Output further oxygenates via the lungs.
SvO2 & SaO2
Disclaimer: The oxygenation capabilities of a patient/animal model lung will depend on the underlying pathophysiology, the level of severity and response to treatment.
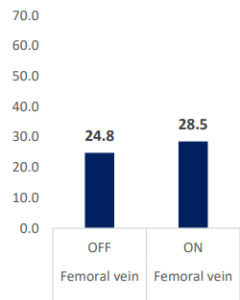
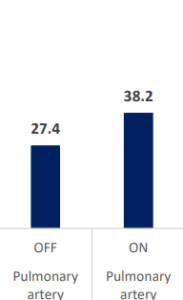
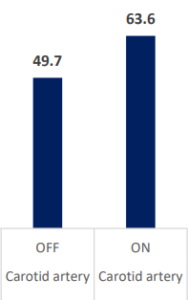
INSPIRA™ ART Provides Significant Increase In PO2 Levels
PaO2 & PvO2 (mmHg)
| Parameter | Blood sample location | INSPIRA ART Status |
INSPIRA ART Status |
Difference | Std Error | t Ratio | Prob>|t| | Lower 95% | Upper 95% |
| PO2 (mmHg) | Femoral vein | OFF | ON | -3.688546 | 1.316849 | -2.8 | 0.0122 | -6.46393 | -0.91316 |
| PO2 (mmHg) | Pulmonary artery | OFF | ON | -10.75 | 1.632462 | -6.59 | <.0001 | -14.1797 | -7.32033 |
| PO2 (mmHg) | Carotid artery | OFF | ON | -13.833333 | 1.272559 | -10.87 | <.0001 | -16.5069 | -11.1598 |
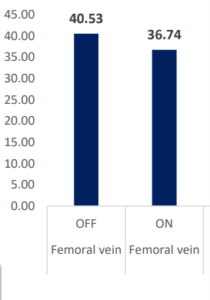
CO2 Removal Via INSPIRA™ ART Is Significant
PaCO2 & PvCO2 (mmHg)
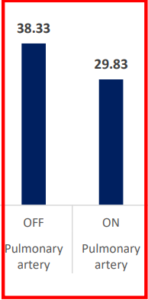
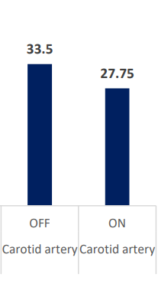
contribution to
PCO2 decrease
| Parameter | Blood sample location | INSPIRA ART Status |
INSPIRA ART Status |
Difference | Std Error | t Ratio | Prob>|t| | Lower 95% | Upper 95% |
| PCO2 (mmHg) | Femoral vein | OFF | ON | 3.8 | 2.591466 | 1.46 | 0.1609 | -1.64929 | 9.220551 |
| PCO2 (mmHg) | Pulmonary artery | OFF | ON | 8.5 | 1.707613 | 4.98 | <.0001 | 4.91244 1 | 12.08756 |
| PCO2 (mmHg) | Carotid artery | OFF | ON | 5.8 | 0.699289 | 8.22 | <.0001 | 4.28085 | 7.219151 |