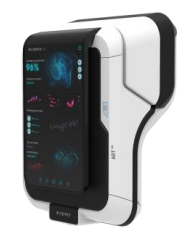
About INSPIRA ART500 System
About INSPIRA™ ART System
Forward Looking Statement
This presentation contains express or implied forward-looking statements pursuant to U.S. Federal securities laws. For example, the Company is using forward looking statements when it discusses the INSPIRA ART’s potential benefits, the INSPIRA ART business model, its vision, advantages of the INSPIRA ART and the advantages of the INSPIRA ART’s components. These forward-looking statements and their implications are based on the current expectations of the
management of the Company only and are subject to a number of factors and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Except as otherwise required by law, the Company undertakes no obligation to publicly release any revisions to these forward-looking statements to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events. More detailed information about the risks and uncertainties affecting the Company is contained under the heading “Risk Factors” in the Company’s Registration Statement on Form F-1 filed with the SEC, which is available on the SEC’s website, www.sec.gov.
About Inspira™
• Minimize the need for mechanical ventilation• An Israeli medical device company, incorporated in 2018
• Mission- to create a paradigm shift in respiratory care
• Multiple patent applications supporting both innovation and intended use
• Supporting initial pre-clinical evidence (In-vivo)
• Management team with track record in medical & public companies
The INSPIRA™ ART System*
● A novel early-stage extracorporeal approach
● Potentially minimize the need for invasive mechanical ventilation (MV)
● Designed to be applied in awake & spontaneously
breathing patients
Business
● The oxygenation and carbon dioxide removal respiratory system substituting MV ,designed to be used both in and beyond the ICU
● US- a primary target market
● Recurring revenue business model based on sale of disposables
* INSPIRA ART system – An extracorporeal respiratory support system composed of an oxygenator, a pump and pump head a plug-and-play cINSPIRA ARTridge, sensors, and a control unit. The Company’s product has not yet been tested or used in humans and has not been approved by the U.S. Food and Drug Administration (FDA) or the CE or other required regulatory agencies.
Vision | Redefining Artificial, Respiration, to impact millions of lives
A New Approach to Minimize the Need for Mechanical Ventilation
Mechanical Ventilation
IV sedation
Intubation
Artificial Respiration
Ventilator Associated Complications
Weaning
Discharge
Inspira’s INSPIRA™ ART System*
Awake Patient
Cannula Insertion
Hemo-dynamic extracorporeal treatment
Discharge
* These photos are for illustration purposes only. The Company’s product has not yet been tested or used in humans and has not been approved by the U.S. Food and Drug Administration (FDA) or the CE or other required regulatory agencies.
About INSPIRA™ ART
Extracorporeal approach
A first-in-class system specifically designed to provide continuous low-flow venous-venous extracorporeal respiratory support
Cannula is designed to draw blood at a rate of 1-1.5L per minute¹
…………………………………………………………………………………………………………………………………………………………………………………………………………………………
Rapid oxygenation improvement & CO2 removal
O2 saturation level is raised in minutes²
…………………………………………………………………………………………………………………………………………………………………………………………………………………………
Low flow utilization
Designed to reduce clotting and hemolysis
…………………………………………………………………………………………………………………………………………………………………………………………………………………………
Compact & portable
Designed for ICUs, general medical wards & ambulatory settings
1. In adult patients2. In August 2020, animal studies were performed in swine model at LAHAV CRO in Israel. 25 hypoxemic events were induced, in order to test INSPIRA ARTs’ oxygenation and carbon dioxide removal capabilities. In 20 out of the 25 hypoxemic events induced, INSPIRA ART treatment was provided with a blood flow rate of 1 liter per minute.
The Cannula

Small diameter
Dual lumen cannula, 16-21Fr in diameter*
One entry point, reduces risks of bleeding and infections
…………………………………………………………………………………………………………………………………………………………………………………………………………………………
Simple placement
Cannula is inserted to the jugular vein and positioned in the
superior vena cava, above the right atrium
…………………………………………………………………………………………………………………………………………………………………………………………………………………………
Reduces the need for medical personnel who are specialized in extracorporeal respiratory systems
Cannula designed to be inserted by any ICU practition** Reduces the need for surgical team
…………………………………………………………………………………………………………………………………………………………………………………………………………………………
Minimize the need for radiography
Designed short length cannula minimize the need for positioning
* The diameter of the cannula depends on the flow volume required to perform the treatment. As the volume of flow decreases, the cannula diameter is reduced accordingly.
** During the market penetration phase, the Company anticipates the INSPIRA ART will be used in ICUs
Designed to Allow Utilization In Hospitals Without Prior ECMO Experience
A novel design minimizes the need for a perfusionist*

A novel “plug and play” disposable cartridge
• Minimize the need for a perfusionist to put together disposable parts
• Any nurse can insert cartridge and operate the system
A Novel auto-priming system
• Priming is done automatically
• Designed to prevent human errors
• Minimize the need for a perfusionist to assemble disposable parts
• Any nurse can insert cartridge and operate the system
* Perfusionist – operates a heart-lung machine (extracorporeal respiratory system), which is an artificial blood pump, which propels oxygenated blood to the patient’s tissues (Britannica.com)
INSPIRA™ ART’s Proprietary HemoProtective Flow Technology Features
Blood protection
| INSPIRA ART’s designed features | Hemolysis reduction | Thrombosis reduction |
| A unique (Low) flow-velocity ratio maintained throughout blood passage in disposable set | V | V |
| A low-volume oxygenator | V | |
| Pump head design for low flow | V | V |
| Short dual lumen cannula design | V | |
| Entire disposable set is coated with anticoagulants | V |
Hemolysis –breakdown or destruction of red blood cells so that the contained oxygen carrying pigment hemoglobin is freed into the surrounding medium²
Thrombosis – formation of a blood clot in the heart or in a blood vessel³
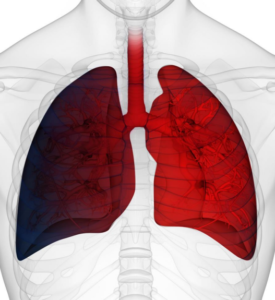
1-1.5 liters of blood are being enriched with oxygen, rebalance patient’s oxygen saturation levels in minutes¹
60+ Seconds to Relief*
“I was able to witness the insertion and positioning of the INSPIRA ART cannula, which was completed without the need for imaging. The INSPIRA ART system’s capability to elevate oxygen saturation levels in a stable fashion, and all within a minute, is outstanding ”.
Prof. Eli Gabbay, Professor of Respiratory Medicine (Australia)
Pre-Clinical Results¹
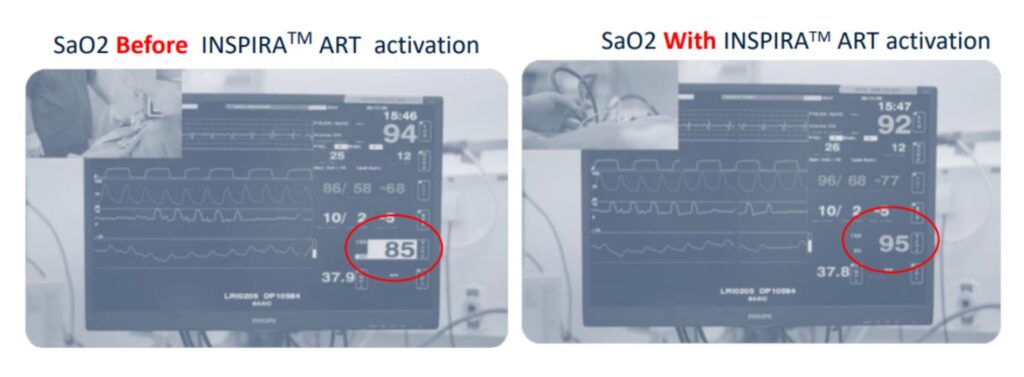
1. In August 2020, animal studies were performed in swine model at LAHAV CRO in Israel